This is a living document, with one section header corresponding to each date with a notable entry. It acts pretty much like my diary for the lab, and honestly, its more for me than it is for you, but if you find it interesting here it is.
I am starting fresh with this new document today, but I may occasionally back port some old entries from my old local diary to fill in some gaps & interesting anecdotes throughout the future.
06-04 - Tuesday
Its been almost a month since my last tempo update. It isn’t because I haven’t been working, its mostly because I’ve been working so hard that I haven’t had time to sit down and write until this last week. I am working on a long-form post describing that work right now, but it will be probably the end of the week before you see it. It will be linked here.
Catching up
So whats happened in the last few weeks? Uhhh… a lot? What was supposed to be a 5-7 day tune up on the SEM turned into a 3 week rebuild from hell after we discovered major oil contamination issues while tearing the column down. We didn’t even really touch the SEM until day 5 of our “SEM work” either, because after running some tests, we found the electromagnetic noise from our chiller (which was also inadequate for cooling our system) to be too much for it to exist within 20 feet of the column. This facilitated a reworking of our entire plumbing setup, which was not a quick and easy task. Our ultrasonic cleaner also kicked the bucket within the first couple hours of cleaning parts, and our entire parts cleaning setup was not nearly at the caliper it needed to be at to clean literally an entire scanning electron microscope. This caused us to have to build our own RO/DI water system to spray HUNDREDS of gallons on parts for rinsing, a new sink and setup for washing these contaminated parts, etc. And we haven’t even gotten to the brunt of what was wrong with the system yet.
Needless to say the system looks like this right now:

You might see here a pile of parts, what I see here is many hours of scrubbing, sonicating, and drying, tons of pieces of this instrument. This table represents nearly 60 hours of work just accounting for the cleaning.
So, the good news is, every one of the pieces wrapped in tinfoil has been vigorously decontaminated and cleaned. The bad news is, our SEM tech Adam, who graciously donated a lot of extra time to us, finally had to leave us for a couple weeks. We will be resuming work in probably 2-3 weeks on this project and finally re-assembling everything. In the mean time, I have a lot of other parts of the lab I need to get in order, and a lot to clean up from this project.
Thats mostly what I have been doing today so far, catching up. I got a nice new eye-wash installed in the lab, did some organization, some returns, some orders, etc. I have a stack of about 60 receipts to enter from the last 3 weeks sitting behind me, and a lot of random stuff that I have been neglecting the last few weeks since this project got so insane. I am going to try to continue to make regular updates as I work and not get out of the habit again, but hey, at least you’re getting a nice long-form post about all that soon.
05-04 - Monday
Acquiring supplies
Today my primary and only goal was to source a water chiller off facebook for some extra firepower for our cooling setup. I managed to get in touch with a guy up in Sturgis last night who had a Vevor CW-5200, a decent industrial chiller, a short drive away. I made a deal to come pick it up this morning.

After grabbing that, I had to also stock up on some helium.

The helium is also to aid our leak checking efforts, since its atoms are so light, it gets sucked into vacuum systems more easily than almost anything else. The idea behind helium leak checking is we attach a device that is extremely sensitive to helium atoms, usually a mass spec tuned just for helium, to the part of the system you want to check. You then spray helium around possible areas of concern, and if any gets through, you’ve found your leak.
Adam is bringing one out later this week, it will be fun to get our system airtight again!
05-03 - Sunday
Today I finally managed to get the leak rate check done. I started by stripping the panels off the main microscope body and taking initial measurements.


Vacuum Leak Rate Check Procedure for JSM-6400
Just for fun, here is the procedure I am using to perform this leak rate check.
Step 1: Pump down and take initial measurements
The very first thing we need to do is leave the SEM running and pumping down in auto vacuum mode for a few days. At this point in the week, it has been on for a week already, so that is done. Once that is done, we need to take some initial measurements on our PG 1, 2, and 3 test points.
Initial Values
| Gauge | Initial Value (μA) |
|---|---|
| PG1 — Chamber | -1.0 |
| PG2 — Gun Chamber | -0.5 |
| PG3 — Buffer Tank | 0.2 |
Our initial values for the PG test points were all between -1μA and 0.2μA, which is odd. It seems like the last person who calibrated this system did so incorrectly, as the set points for these gauges should not be 0.
Step 2: Stage switches for correct valving
Next we need to set the switches on the front panel of the SEM manually to close all of the valves. To do this we first ensure our AUTO/MANUAL switch is still set to AUTO, and then flip every switch to its down/off position except for the RP and DP switches, to keep our rotary pump and diffusion pumps running.


Step 3: Flip to manual and measure values
I would re-show the picture but its probably the last picture you looked at, we are now going to flip the AUTO/MANUAL switch to MANUAL.
Once we flip to manual we QUICKLY check our PG3 test point and look for a sudden increase, if we were to see a sudden increase, we would want to open V2 to re-up the backing pressure on the diffusion pumps.

Bearing all this in mind, we flip the system to MANUAL. This produces quite a lot of noise, from all of the valves closing at once and the system quickly flashes an air pressure warning before it goes away, but otherwise, everything is quiet.
I quickly measure PG3 first, and notice that somehow it has actually decreased… ok? That is either a really good sign or really bad sign. Down from 0.2μA to 0.1μA. Regardless, it doesn’t show a sudden rise, so I leave V2 closed for now.
I take a quick round of measurements at 15 seconds after confirming PG3 is ok, and then start repeating this process every 60 seconds to start to gather data.

At this point we are seeing a big leak in the chamber at the very minimum. The gun chamber also shows a leak, but definitely a slower one, and the buffer tank / PG3 is still just kinda… sitting there suspiciously?
I continue to collect data for the next 45 minutes. At the 22 minute mark we actually see PG3 jump up from 0.1μA to 0.2μA. All of the other test points have changed far more. I start deliberating with some members of the SEM discord, and Adam and I decide it might be a good idea to open V2, just in case. We expect to see the value come back down if the meter is working in any regard, but to see this little degree of change over this large of an amount of time is concerning. It either means the vacuum is actually really good, or something is really wrong down in the buffer tank.
After 38 min I open V2 to pull backing pressure back onto the buffer tank and diff pumps, and sure enough, one minute later, the value climbed back down to 0.1μA.

So we know PG3 is either fine or completely toasted, what about the other two? Well, lets take a look at a graph here:
We can see pretty clearly that we have a bigggg leak on PG1, our chamber. Its such a massive leak in fact, that it severely skews the scaling of the other two test points. Lets flip this graph’s y axis to a log scale so we can make better sense of the smaller values:
Ok this makes it pretty clear to see whats going on with all 3 PG test points over the course of the 45 min run. We have identified our Chamber has what could basically be described as a gaping hole in it in vacuum terms, our gun chamber is not winning any awards, and our buffer tank has either a goddamn perfect vacuum or some weird stuff going on. This gives us a lot of background data and lets us know where to start focusing when Adam gets here later this week. Its likely at this leak rate that the big issue with the chamber door is probably a fiber across an O-ring. When Adam has his helium leak detector out here later this week, it will be easier to figure out whats going on.
Other
Tonight my only other goal is to source an extra water chiller for the lab in the next day. I am going to hit facebook marketplace and get back to you guys with an update tomorrow.
05-01 - Friday
Lighter day today, I am leaving a bit early since I am planning on working an ungodly amount of hours next week with Adam on the SEM. Today I have mostly been cleaning up and organizing the lab / continuing to get ready for Tuesday. If Adam gets back to me with the leak check procedure Ill do that today otherwise Ill leave that for tomorrow evening or something.
One of our interns and I also spent some of this afternoon organizing some cables to get our SEM inventory completed ahead of Adam’s arrival next week.
04-30 - Thursday
I was going to start today by performing the manual leak check on the SEM, but I still haven’t been able to confirm the valving order with the experts on this machine yet. I am thinking about re-starting this pump down on Saturday and just making this order of business #1 when Adam gets out here. But we will see, I sent some messages and if I don’t hear back today I’ll likely just postpone this process until next week.
General
I spent a lot of this morning trying to track down the power supplies for some PTZ Optics cameras we have, considering trying to use these for our live-streaming next week. No luck on that but I am going to order some replacements that should be here Tuesday.
I had a call with Adam McCombs earlier today and they gave me some good advice on leak checking, I am still waiting on them to confirm some stuff around valving, but we confirmed all the details for their visit out next week, and we should be in for quite a treat. It will be really fun to document this process, and I am excited to update all of you lots on that next week.
Most of my day today was spent just trying to figure out logistics for next week, and I also spent some time with Esteban and Bri today getting Esteban introduced to the lab for his new phylogenetic research.
04-29 - Wednesday
SEM Updates
I started today with a tool run to Harbor Freight. I needed a new ųA meter for testing the PiG 1, 2, and 3 test points on the SEM’s vacuum board. The SEM has been running for a couple days because we are going to run a leak checking procedure tomorrow to get a good idea of where the vac system is at before Adam gets here next week:

After getting back form Harbor Freight, I did a quick probe of the test points to see where we were at. The main PiG 1 test point was bouncing between 0-1.4 ųA which I am guessing means that gauge is at the lower end of its range in being able to detect a vacuum, so that vacuum is probably pretty good right now. Tomorrow we will manually shut a few valves and measure the rate at which that value climbs across a couple of those points, and that will give us a leak rate.
I forgot to take pictures until i was putting the last screws back in, but here is a peak into the vacuum test panel:

Sputter Coater Updates
Our Global O-Ring & Seal order got here today, and our amazon order got here a couple days ago, so we now have all, or at least nearly all of the vacuum hardware that we will need to get the sputter coater back to a full vacuum.

I will update the sputter coater log when I actually test fit these, but for today I was just taking inventory of what we received.
04-28 - Tuesday
First update in a bit here, I was out last week for some personal stuff and was out sick yesterday. Happy to finally be back.
First exciting update for the day is that we finally got Adam McCombs scheduled to come out and help us fix our SEM up next week. So the 4th-9th-ish will be full of many exciting SEM updates getting that system ready to use for our interns this summer.
Secondly, I had a great chat with my buddy Esteban this morning, who is going to start doing an OpenIDI research project on some potentially new trilliums.
Today I am mostly just getting caught up from a few days of being out. I am taking inventory on some of the SEM issues, and trying to build a plan to get the sputter coater and all parts of the SEM workflow fully operational before June.
I also made some quick code fixes to this blog, namely with how sidebar scrolling works when sidebar content gets long.
04-22 - Wednesday
Today my work has once again mostly been on the sputter coater, I spent the day sourcing and ordering replacement o-rings, trying a few different cleaning methodologies, and planning some future parts of the project. You can read about all that here: Sputter Coater Repair Log.
04-21 - Tuesday
Today I am doing some more work on the sputter coater, see the sputter coater repair log post for more info on that: Sputter Coater Repair Log. A lot of what I have been working on today is in that doc, and today’s tempo is probably going to be a bit lighter due to that and an appt I need to make this afternoon.
I am taking part of the afternoon here to work through re-reading this paper, to fill in some gaps in my understanding with some of our current gall research. I read it months ago, but it again has been months and I need a good refresher.
I also spent some time today fixing more bugs on this blog codebase & getting some behavior with anchor links and some css resolved that my agent was having a hard time with.
04-20 - Monday
Today we can finally re-organize the lab! Now that the BSC is up on wheels it should be easy to get all the equipment moved to where it needs to go.
Lab Organization Update
Bri and I spent a couple hours re-organizing everything this morning:

After a few hours we have it in a really good spot:
This is great, because it lets us have both of our freezers as well as the autoclave all contained within the wetlab now, which is a huge gamechanger for our workflows.
Having this -86C up all the time also enables us to do a lot more work with sensitive peptides / DNA. I am going to email Adam Kranz this afternoon and tell him we are likely ready to recieve the peptides.
Since we had to move some of our art around, I opted to quickly replace it with some posters that are more relevant to our research:


Gall research
Speaking of our research, I also reached out to one of our research partners; Adam Kranz, today, and told him we are ready to start Phase 1 of the Arabidopsis trials in earnest. We will hopefully hop on a call soon and figure out what next steps look like.
I’ve also spent quite a while reading around on “The Levin Lab” today, and going through some course materials there, just to further my own knowledge. Learning is fun 😉. Speaking of learning, I have a biology textbook that just got delivered to my house, so I am going to sign off for the day and dig into that. See y’all tomorrow!
04-17 - Friday
No update yesterday because it was mostly an amp lab day. Yesterday evening / this morning though I made some progress on the Echinocereus OpenIDI project.
Echinocereus Spine Imaging
Today I managed to get some imaging done of the first specimen that I prepped using St. Francis’ sputter coater last month. This specimen is Echinocereus relictus or Relict Hedgehog Cactus. This imaging is part of a larger study to characterize members of the genus based off of the morphology of these tiny spines. We got a few good images this time:



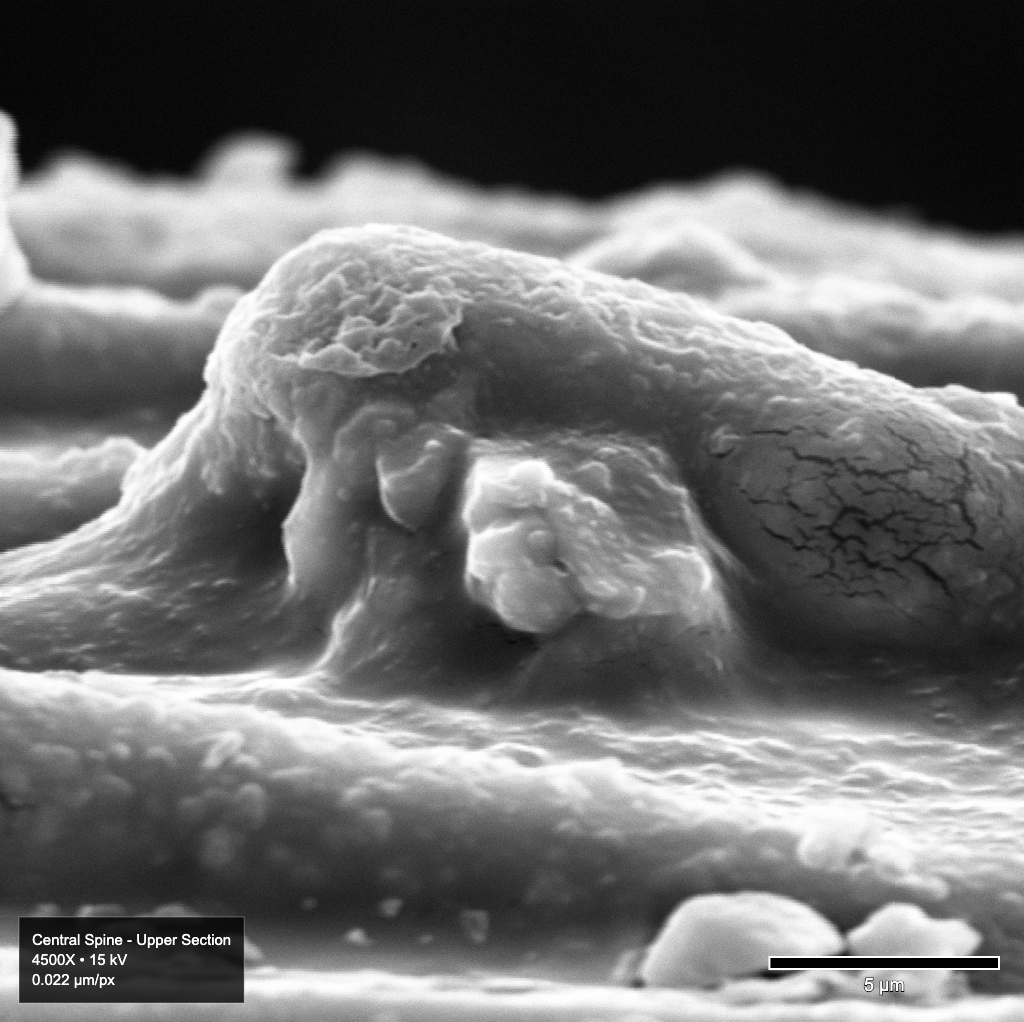
These turned out pretty well. I am still getting some flagging on some of these, but once I get these electrical issues troubleshooted properly, I think that will all go away. Plus, its at such a small scale it doesn’t impede the research we are doing for this study. To any interested readers, full uncompressed images are viewable in our SEM image viewer tool on OpenIDI. Simply scroll to the bottom of the page for this specimen to explore the full detail images and zoom to your heart’s content.
My main images with the SEM at this point are the flagging like we see in these images. I am going to attempt to measure electric fields in the room next time we are running and see if I can pin down the source of the electrical noise. Fixing this will enable higher resolution scanning and more consistent results.
BSC
In what is hopefully my final update on the BSC, we FINALLY got it up on its new casters today, and I am happy to report it wheels around happily without complaint.


With this back down and fully moveable with just one human arm, we are poised to be able to re-organize the lab pretty easily on Monday!
Other
I also did a bunch of code cleanup on the raw SEM Image viewer this morning, now as an admin I can re-label / correct meta that was labeled incorrectly on the SEM computer. I used it already to help me fix these new images with the correct labels.
04-15 - Wednesday
I got in today after some appts and checked on the time-lapse I started yesterday. The focus held up a lot better this time, and this gives me a lot more hope for managing these longer time-lapses. We did end up having a lot of extra microbial growth visible on the slide this time, we are still getting blooms of bacteria and mold after 48ish hours. This is a slight problem, but one I think we will be able to alleviate through better sterilization of our soils and better sterile conditions when prepping slides. I am also considering adding some sort of anti-microbial agent to the vitamin solution we add to the slides.
Its pretty interesting seeing the ‘flow’ of the microbes crashing into this root tip wave after wave. You can see the image darken about 1/4 through the time-lapse, this is because I turned down the gain on the transmitted light detector because I noticed the microbes were sort of stealing the show. I don’t think this time-lapse holds a ton of scientific value in terms of what it teaches us for our study, but its a good baseline to try to beat for keeping the sample microbe free and clearly imaging.
I am going to get another one started first thing in the morning, and I have some cool new ideas for this one including using PFS from the start, and I also want to dig a bit into the confocal’s spectral analysis tools to see if we can’t get a better idea of what is fluorescing in our samples.
04-14 - Tuesday
Today and tomorrow I have some doctors appts, so updates will be a little sparse.
Arabidopsis Update
The time-lapse we started yesterday actually looks great, though, the root pulled out of focus about halfway through:
I got another timelapse started today with the same sample, I used the scope’s PFS (Perfect Focus System) to keep the focus plane in line with our subject this time, so we will see how that all turns out tomorrow.
I also spent a while looking into some plant autofluorescence and trying to build some hypotheses about what we are seeing in our samples.
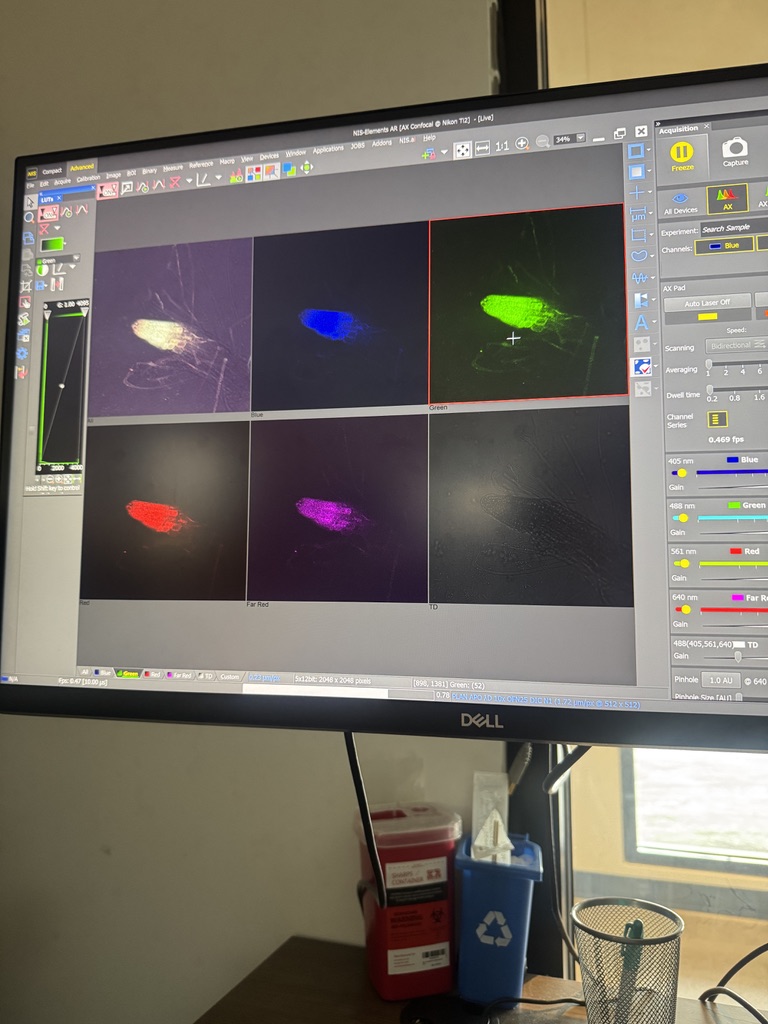

I am going to let this timelapse cook and check it after my appts today and tomorrow!
04-13 - Monday
Got rolling this week by attempting to re-organize the lab again and get the BSC situated finally. We borrrowed a lift from DoItBest and were able to get the BSC lifted up, take off the broken casters, and set it back down.

After the BSC was moved, I poured some new plates to use in Arabidopsis imaging. I am doing this because I am just about out, and during the prep for my next time-lapse here in a bit, I will use the last of my supply.

Once the new plates were ready to go, I went ahead and prepped one of the plants for imaging. This is a fairly simple process (that frankly I should have gotten more pictures of) where we take one of the small individuals that has been growing, clean all the dirt off of its roots, place it in a small petri dish, add a vitamin mixture, and then sandwich a cut-out of fresh agar from one of the aforementioned plates onto the roots to hold it in place and keep it hydrated. This is how we have been preparing our samples thus far, but I think next time we are going to make a small modification and cut the agar around the leaves to provide them with more oxygen.
After prepping, I loaded it up onto the scope and started the image:


In these last few pre-experiment trials, I am trying to push our time-lapses longer and longer, and start to lock in on final laser settings.
04-10 - Friday
Housekeeping, made some updates to the Sputter Coater Repair Log, did some minor code work and tweaks to this site, and am working on doing some expenses and other random admin work. We have to wait to move the BSC until Monday because the lift isn’t quite ready for us to borrow yet.
04-09 - Thursday
Today I was helping out down in amp lab, so I don’t have too many updates on the regular projects. I probably wouldn’t have even posted today, but I got the freezer fixed and wanted to show it off
Freezer update
The new plug came in today, so I took a little bit to get it swapped in and get things re-assembled!


Random
I also ran a test print with our new top loading filament extruder setup, and it worked very well. This should allow us to print most weird filaments. I did a test benchy and it came out great.


04-08 - Wednesday
More freezer stuff
Last night I left the new freezer running to see if it would get to its set point, this morning I was pleasantly surprised to see that it was there. Now that I know it works, I need to get it moved to a more convenient spot so its not getting in the way for people that actually need to use the lab today. I am going to unplug it and wheel it over into the imaging lab for now, Bri and I should hopefully be attempting to fix the BSC (BioSafety Cabinet) positioning today, which should free up enough space to let this freezer have a proper home.


Code stuff
I spent a couple hours this morning making some improvements to this blog codebase for our other editors here at BNX. I got Cameron set up with the new tooling, I’m sure you’ll see a post by him here shortly. I also made some optimizations surrounding image loading and sizing. I think the new setup will be a much better experience for content creators and readers alike.
Other
I spent a while today working with one of our interns to try to figure out how to get our Elveflow microfluidics system talking to our Nikon Ti2. It seems like we could basically get it working in a few minutes if we had the TTL/Analog IO module for the Ti2, which we do not. We are reaching out to Nikon to get a price on this module. Hopefully its reasonable and we are able to make it work, because we will need TTL capabilities for a lot of our experiments in the future.
Bri and I got confirmation that DoItBest is going to let us borrow their lift tomorrow so we can fix the BSC positioning, so tomorrow should be fun 😛
04-07 - Tuesday
Today I am working on checking the state of all of the new equipment we got yesterday.
Freezer work
I started by pulling the freezer into the wet lab so I could power it and test it. As soon as I went to plug it in, I noticed that the plug didn’t go in at all. After some investigating, I found that it arrived to us with a broken plug. No bueno.

I stripped the side panels off and made my way into the power supply, and was able to pull the plug out from the back so I could easily read its model number:


After some digging I found it to be a Qualtek 744W-00/04:
I got a replacement unit ordered which should be here this week. In the interim I will likely just wire it up for a quick test.
I got the freezer plugged in a totally-safe-and-not-jank way for testing:

Luckily, it booted right up. We ended up having a proper dedicated (at least we are fairly sure its dedicated & are checking with our electrical contractors) outlet in the lab already for this, and its compressors booted right up without tripping anything, so assuming its good for now.


I am going to leave it running overnight, and see if we are down to near the -86 C range by the morning
Housekeeping
Now that we have this new freezer, we need to re-organize the lab to actually fit it. Right now, this is very difficult due to our friend “The BioSafety Cabinet”. Due to an unfortunate situation with its casters, its incredibly hard to move right now.


This is a big problem because now the only way we can properly fit all of our equipment into this lab where it needs to go is by moving this BSC. Bri and I have been brainstorming ways to lift it by its frame all afternoon, and I think we have some ideas. You will probably see another update on that soon, just… not today.
Random
I am also setting up Parallels today because I was recommended Solid Edge for a few of my projects and I want to try it out. I’ve also been printing some things to finish our printers top feeing AMS HT setup.
04-06 - Monday
Today Bri and I went to Indy to pick up our 3 new vacuum pumps and new -86 C freezer. There isn’t much to say about today other than that it went off totally without a hitch. One of the easiest equipment moves we have done since we started moving heavy equipment into this lab. I think we are getting the hang of it 😎
Here is a photodump from today:



04-03 - Friday
Housekeeping
Spent this morning finalizing our GovDeals order & Tax Free status, got a truck rented for Monday so we can go pick up our new goodies. Printed more adapters for the 3D printer and new AMS mounts.
This morning I’ve been doing a lot more research on ways we can schedule scope time. I am considering building it into our existing lab software, nucleus.
I spent most of the day doing random admin work and testing out a few ideas / concepts in code. I am leaning towards quickly building our own scheduling suite for our lab tools into our site.
04-02 - Thursday
Printer Upgrades
Started the day off with finishing some organization of our 3D printer station. This print finally finished so I spent some time getting the station all organized and looking nice.

Before we had a rack that held both of the AMS units on top of the printer. This worked fine, but made using the laser and a top-loading extruder configuration very difficult. This will solve those problems and give us a lot more flexibility with how we use this machine.
We also swapped nozzles to fresh ones, and fixed the nozzle blocker which had broken.
General
I fixed some tax exemption stuff for GovDeals, hopefully they get back in the next day and we can finish paying for our lots.
Arabidopsis


Decided to spend some time today starting to get another time-lapse prepared on the confocal scope. I got a new plate poured with some fresh Agar, spent a while getting the sample prepped, and then realized that the BioFilm Group was already using our scope 😭. I fridged the samples, hopefully they will still be ready to image in 46 hours when the group’s z-stack is done. If not I’ll re-prep Monday.
04-01 - Wednesday
Today I spent the first part of my morning babysitting some auctions for new equipment as well as figuring out tax exempt status for the auction house. I am happy to report we won 3 very nice vacuum pumps at a nice price, as well as a -86 C freezer which will come in handy with our DNA work. We will likely go pick those up on Monday.
I also got the rotator re-built yet again (for the last time, if it breaks again we are officially buying a new one). It seems to be real nice an quiet after applying the proper grease (shocker 😲)
Sounding real nice good.
03-31 - Tuesday
The oil got delayed another day somehow, so I guess I will not be fixing the auto-rotator today. It will have to wait until tomorrow. This morning so far has just been the repeated process of beginning to start work on a project before realizing shortly after that I don’t have the tools, and thus switching to another project and repeating.
I spent a while troubleshooting issues with our 3D printer, and decided to print a new rack for our dual AMS setup so that we can more easily access the top of the printer.
The water hose I ordered for the sputter coater also came in today, a test fit around the barbs worked quite nicely, and I have no doubts it will fit correctly.
03-30 - Monday
Ok so the grease I had on hand was also not the move, the gearbox was audible from the hallway this morning which is not amazing. Luckily the grease I ordered Friday is going to be here today and should be more or less exactly what we need. I got this rotator for incredibly cheap, but if this grease replacement fails on me one more time I am going to just be ordering a new one and scrapping this one for its internals.
I have a few projects I really want to get done by the end of April:
- Adam McCombs is coming to visit on the 12th of April for a whole week, we are going to be doing an intense SEM tune up. I need to make sure the lab is clean and we have all the tools we need to work on it that week.
- I want to get the sputter coater fixed
- I want to image every spine from the Echinocereus project
Most of what I am working on these next few weeks will be related to these projects. Getting the SEM lab fully operational by end of April will help not only my but many of our research associates research.
I ordered some new hoses today for the argon and water lines on the sputter coater system and updated my log accordingly. Took a while to source and confirm the available hoses and find decent deals on all of the diameters I needed, but they are ordered now.
I also got on govdeals and bid on a new vacuum pump for that machine, as well as a new -86 C freezer. Both bids end Wednesday and I hope to secure both of them.
I spent a while running the SEM and just double checking some things anticipating the arrival of Adam in a couple weeks.
Its now 3:37pm and the oil for the gearbox still isn’t here so its likely our Autorotator is down for the count until tomorrow. If it gets here before 4pm I probably have time to get it fixed today, but I am not optimistic.
03-27 - Friday
While I was away
It’s my first day back in a couple weeks (aside from coming in to do some quick maintenance between trips last week). I saw and did a lot of really cool things while I was out and about, including potentially getting an international partnership started with some OpenIDI stuff, and also filmed a documentary about some ecological threats facing the Big Bend region that I hope to finish and release soon. The way I look at it if im traveling to a new place and not trying to build ecological relationships, I am not doing it right 😅
Back to the lab
I was greeted walking in this morning by the sound of our variable auto-rotator grinding its own gearbox into a fine metallic paste. It needs new oil very badly. We re-oiled it a month ago, but the red-n-tacky grease we stole from the robotics lab is probably not the right call for something that needs to rotate for hundreds of hours a week. So I need to fix that, I also need to get some plans finalized for some SEM maintenance we have coming up. Start ordering o-rings for the sputter coater, and generally just try to get the SEM lab to a great place by the end of next month. The lines are still pink on the SEM, indicating that the PH is still a little too low, I may swap to a different coolant soon.
Fixing the auto-rotator
For reference, the reason I need to strip this down again is to fix this sound: Click play to subject yourself to a horrible noise
This thing is a little annoying to get apart due to using like 4 different screw standards, but it comes apart quick. I first stripped off the top rotator plate.

I yoinked the top assembly from the machine and threw it in the fridge to cool down. It was a little too hot from being on for about 2 weeks.


After 20 min in the fridge, I pulled the gearbox off the mounting plates and pulled it apart.


Let me tell you, red-n-tacky grease was not the move, look at this horror show:



Sounds much more like a machine running and much less like a gearbox eating itself
With that back together, its sounding OK now.
Other stuff
The most of the rest of my day here I am just catching up on everything from being out and planning out my next few days.
03-05 / 03-06 - Thursday / Friday
Mostly worked on getting the lab cleaned up and organized for my departure, and worked a bit on the SBRI site a bit and got that to a decent starting point.(Summit Biofilm Research Institute, one of our research partners). Nothing really worth adding to a tempo doc, but I will link that site here once it is finally live.
The next couple weeks I am going to be traveling around Jamaica and West Texas, and I hope to come back from these trips with some new leads for OpenIDI. If I research anything interesting during my travels, you can be sure I will add it here.
03-04 - Wednesday
Pink is the new translucent
Today I got into the lab, and I noticed that the coolant I had installed yesterday was pink?


This actually happened last year, I posted in the SEM discord at the time and our general consensus was that it was probably just dye from the old coolant that had run in this system (a pink antifreeze) that had been baked into the lines and coaxed back out by the new coolant. Last year this went away within a few weeks and my lines were back to clear. Now with this happening again a year later, I am not so sure I was correct, but we will see.
I decided to check the pH of the coolant to see if it was maybe super acidic or something and actively corroding the lines. I used a pH strip I had lying around and it was definitely extremely basic, this checks out, as SDS specifies a pH of roughly 11.5.


I checked the both the fresh OptiShield that is unused, and the spent OptiShield, which both at least above 8 (the high end of my test).

I have been conversing with some members of the SEM Discord about these problems today, and a couple users suggested it might be an indicator dye indicating that the PH has dropped into the 8 range. This would make sense, especially when I think back to how last time it slowly changed back to clear over a few weeks. That could have been the indicator dye slowly dissipating as the PH rose back into the 11 range. I am going to pull a cup out and check to see if hitting it with a stronger acid pulls the dye into its <8.3 range color, which should be translucent (if it is a fluorescent dye). A user by the name of TheServer201 shared this graphic:
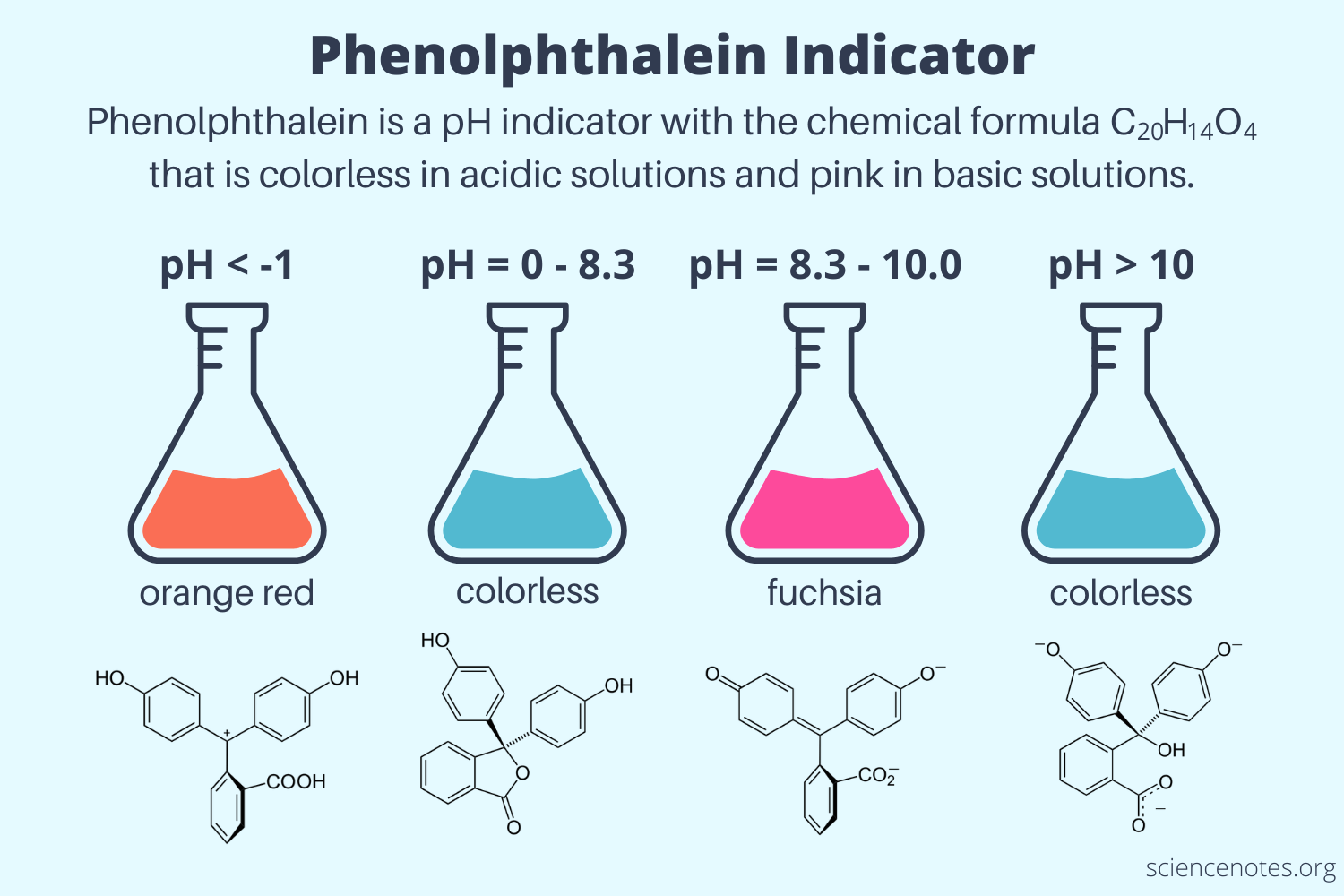
If this were a Phenolphtalein Indicator, its likely that when we add the coolant to the SEM initially, the metals in the system briefly (for a period of a week or two) bring the pH of the solution closer to the 9-10 range than the 11.5 that the fresh stuff is mixed at. This pulls the indicator dye into its fuchsia range, which exists between 8.3 and 10. When we add pure acetic acid to the solution, if it was indicator dye, it would immediately drop into the 0-8.3 range, which should be colorless again.
Lets pour some acetic acid into the coolant and see what happens:
Upon dumping some acetic acid into the coolant we pulled out it cleared up immediately.
It immediately cleared up upon the addition of the smallest drop of acetic acid. To me this proves that this is some sort of indicator dye. I am for now going to file this under “slightly weird but probably not concerning” for now, future opinion subject to change 😛
The rest of the day I just have some code work planned, and I am also preparing to leave for a trip soon, so I may not have an update on this project for a bit.
03-03 - Tuesday
Coolant Swap for the SEM (finally)
I was a couple days behind where I wanted to be with this today because I was feeling sick yesterday. Back at it today, and getting the SEM back up and running.

When I first used my fancy new handle to crack the drain valve, I was immediately met with the sheen of a thin layer of oil which must have been sitting at the bottom of the pump’s drain line for a very long time.

I don’t feel so great about this, but I am inclined to leave it be for now. There aren’t a lot of ways oil possibly could have even gotten into this system. The only way I can think would be some seal failing in the water pump itself, but there are no pathways inside of the SEM or DV-502 which are both hooked to this that would allow oil to migrate into the lines. So it has to be something with the water pump. Its likely I will perform another coolant swap soon to see if we are still pooling oil.
Regardless, I decided to carry on with the change. I pulled a few pans of this mostly coolant with some interspersed oil into a waste bucket:

After pulling a few of those pans out to the point where the reservoir was mostly empty, I took a heavily gloved hand in there with some paper towels and cleaned residue off of the edges and sides of the plastic container that acts as the main reservoir.

I then refilled that reservoir with a flushing dose of our coolant, maybe a gallon and a half, let it run for a bit, drained it again, washed the internal filter down in hot water, and then topped the pump up to its final dose and put it all back together.

After it was all topped off to its final level, I put the equipment corner back together and ran the SEM for a few hours to circulate the coolant.

This was all I had time for today, but it was nice getting it all back up and running.
03-02 - Monday
Got the buckets
I started my morning by running out to menards and grabbing a couple 5 gallon buckets with lids so that I am well-stocked up for coolant swaps in the future. The coolant swap is ready to do, but I am not going to get to it until this afternoon because I have a project this morning that I need to get rolling on first.
OpenIDI Work
One of the projects we are working on right now in the OpenIDI realm is examining the micro-structures of a few Echinocereus sp. under SEM to determine if there are microscopic morphological features that might help us tell certain species apart, that right now, are too hard to tell apart without DNA work. Since our SEM lab is under maintenance, Professor Warreny Pryor at St. Francis University here in town has graciously offered me the use of their SEM and sputter coater. My goal this morning is to get as many of these specimens as I can gold coated.
At the imaging lab
I spent the entire first part of my day over in St. Francis’ imaging lab. We didn’t end up getting any time in on their JEOL NeoScope but I did spend an hour or so prepping and coating 10 sets of echinocereus spines for imaging.


Their coater surprised me actually, its similar to our BioRad Coater that we are currently repairing (read more about that here), but instead of using Argon gas, it just uses whatever was left in the atmosphere after roughing to make its plasma. An interesting and mechanically simple design, but one that does leave your samples more oxidized than they would be had they received this same coating with an atmosphere of inert gas.


After about an hour of prepping and coating samples, I finally had 10 that were ready to image for this project. I coated them for durations ranging from 1-4 mintues. These 10 I have quite a lot of extra material from, so I don’t mind doing some testing on this material. I’ve notated which samples were coated for how much time, so that when I get to imaging I can start to nail down the best coating time for the rest of this experiment.

After my brief stint at St. Francis, I ended up developing a pretty bad migraine and ended my day a bit early today, coolant swap to come tomorrow.
02-27 - Friday
Coolant Swaps and Electrical Work
Today is mostly a maintenance day in the lab. I got in early today to let our electricians at Votaw in for the 4th day in a row. Votaw has been re-working our electrical system to offer more surge protection for our sensitive devices at the breaker-level, and adding more outlets and amperage in the lab so I don’t keep taking out the entire circuit with our High Vacuum Evaporator. They have been overall very pleasant to work with, and I would recommend their services.
Time for a coolant swap
I got started today by preparing to do a coolant swap on the SEM. It needs its coolant swapped because that coolant is about to experience its first birthday, which our fancy sticker here very explicitly warns against:

Honestly, I’m glad for the sticker, because I forgot I only need to flush ~1.5 gallons, I somehow had it in my head that there were 5 gallons of coolant in here. I already have a 5 gallon jug of pre-diluted OptiShield sitting in the lab right now, so I am all set probably for next year’s swap as well.
Learning from mistakes
Last time I emptied this thing, it was mostly distilled water I was running in the loop, which is a lot less nasty to get on your hands than the OptiShield that is in there now. I spilled so. much. water. on the floor. I spilled so much last time for 2 reasons:
- Our oil pan was not perfectly flush against the back of the chiller, I cut a hole in it for the chiller drain spout to fit through almost a year ago, but the ridge still caused the pan to sit out from the drain plug a bit.
- Last time, I had to open this drain valve with a pair of pliers, which slipped a lot, mangled the plastic a bit, and overall was very difficult to quickly open and shut.
I fixed the first issue by trimming the back rim of the oil pan down so that it is now flush with the back of the chiller.
For the valve issue, I quickly whipped up a new design in fusion 360 that acts as a turn handle for that valve.


After 1 quick revision to make the length that grabs the valve stem a bit longer, and the knurls just a touch deeper, this tool is ready to help us open and shut this valve with record ease and speed.

Luckily, I had to disconnect and move all our pumps for the electricians to do work in our space earlier this week anyways, which would normally also be part of this process, so perfect timing on that. I took some extra time to re-position some equipment and give myself a comfortable and ideal work area for this coolant swap.

As always happens when I reach this phase in a project, I realized I don’t have the last thing I need to actually start it, a 5 gallon bucket. So I need to go run to Menards to get one. Its questionable at this point in the day if I will be back in time to do a full coolant swap and re-assembly of the equipment corner this evening, so likely the next update in this doc will be on Monday or if I get some time this weekend, we will see. I am not worried that leaving coolant un-circulating for 2 days past its expiry will damage anything, we just definitely need to get it swapped out before the next time we power it all on.
Code work
On another note, I’ve done a lot of code work today both on this blog and the site at large, cleaning things up and getting Bri set up with an editor so she can start adding some content to this site as well.